Hydrolysed proteins, chitin, and hygienised insect frass should be granted end point status for use in EU Fertilising Products under Regulation (EU) 2019/1009 – EBIC

Eurovent publica la versión actualizada de la Recomentación 14/6 para aparatos de refrigeración - Revista Climaeficiencia | Noticias de Climatización y ACS
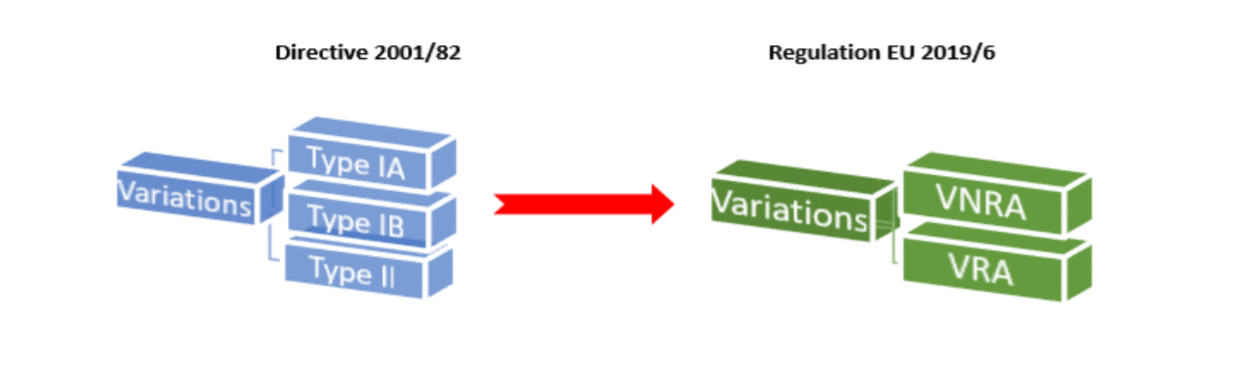
EMA scientific recommendations on delegated and implementing acts as part of the implementation of the new veterinary medicines

Italy | Regulation (EU) 2019/6: Implementing acts on good pharmacovigilance practice for veterinary medicinal products and on the format and content of the reference dossier of the pharmacovigilance system